Dr. .... ......... | DD.MM.YYYY
Patient Summary
MG is an 87 years old female with a history of Hypertension, CAD, GERD, Glaucoma, Mild Pulmonary Fibrosis, and Vertigo. She was diagnosed with metastatic urothelial carcinoma (mets to lungs and bones) and underwent a right radical nephrectomy.
Research Inquiries
- Is there any chemo or immunotherapy regimen that could be recommended to this patient given her advanced age and relative frailty? The intent would be palliative, but the goal is to extend life with a meaningful quality of life (as little toxicity as possible with the least amount of side effects).
- Are there any study protocols/trials or experimental treatments that would meet the above criteria?
Conclusion
Our research focused on three primary therapeutic approaches recommended in the NCCN and European guidelines – chemotherapy, immunotherapy, and radiotherapy – all can have negative impacts on patient quality of life due to long treatment cycles, intravenous administration, and side effects.
However, the reviewed literature and other studies[1] indicate that immune checkpoint inhibitors may be better tolerated in elderly patients compared to chemotherapy in terms of quality of life and toxicity. Immunotherapy, specifically Pembrolizumab and Atezolizumab, have shown extended median overall survival when the tumor expresses the appropriate genetic criteria. Additionally, anti-FGFR therapy, such as Erdafitinib, is a potential treatment option when considering QOL aspects for patients whose tumor expresses GFR2/3 mutations or FGFR3 fusions, as it can be given orally.
Radiotherapy can be considered to relieve local urothelial symptoms, but it didn’t show to have better survival outcomes. If chemotherapy still needs to be considered, Carboplatin is considered less toxic than Cisplatin in the GC treatment regime (Cisplatin\Carboplatin and Gemcitabine)[15], and the GC treatment regime is considered less toxic than methotrexate, vinblastine, doxorubicin, and cisplatin (dd-MVAC) regime.
Regarding clinical trials, we included three trials that may be relevant to the patient. However, considering the hundreds of active clinical trials for urothelial carcinoma in the US and Canada, more information about the patient’s medical history, medications, tumor mutations, and personal preferences is necessary to determine if the trials are suitable for her clinical status and personal preferences.
Important Note
Neither the services nor the research report constitute medical advice of any kind and are not intended to be a substitute for professional medical advice.
Medical Meta Findings
- Is there any chemo or immunotherapy regimen that could be recommended to this patient given her advanced age and relative frailty?
Data summarizing the most up-to-date common practice treatment work-flow regarding metastatic Urothelial cancer – according to the National Comprehensive Cancer Network Bladder Cance guidelines[2] published in 2023, metastatic urinary tract disease is worked up by the following scheme:

The only interventional palliative therapy mentioned in the guidelines is radiotherapy. In the case of systemic therapy, the guidelines mentioned chemotherapy and immunotherapy:
Chemotherapy:
According to the European Association of Urology guidelines[3] for Upper Urinary Tract Urothelial Cell Carcinoma (UTUC) published in 2023, Upper tract UC and urothelial BC both respond to systemic platinum-based chemotherapy. Therefore, cisplatin-containing combination chemotherapy is the standard treatment for advanced or metastatic UTUC:
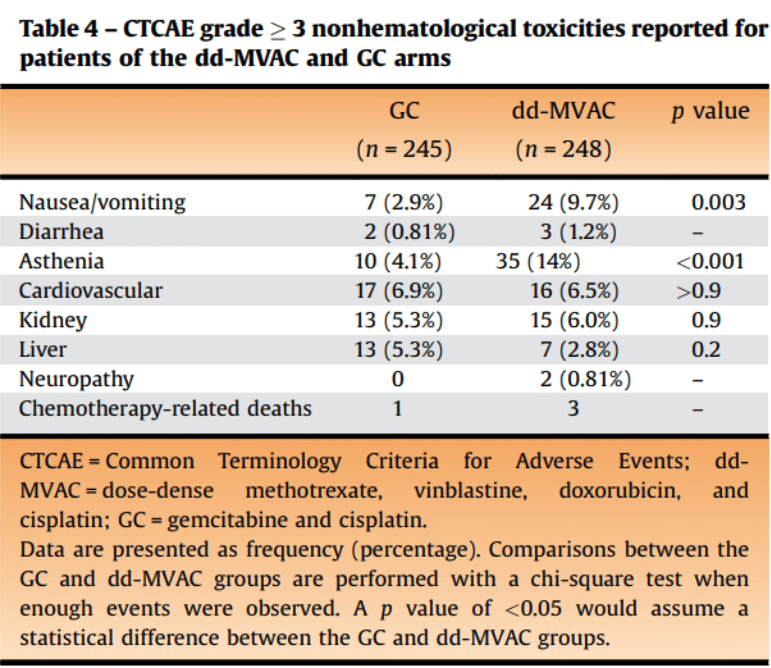
First-line gemcitabine and cisplatin (GC) treatment regime is less toxic than methotrexate, vinblastine, doxorubicin, and cisplatin (dd-MVAC) regime – an article[4] published in European Urology journal (Q1, impact factor 24.344) in 2021 shown the secondary outcomes results of phase III clinical trial comparing the two first-line IV chemotherapy regimes in 500 patients with urothelial and bladder cancer. According to the results, most of the Common Terminology Criteria for Adverse Events (CTCAE) grade 3 toxicities concerned hematological toxicities, reported for 129 (52%) patients in the dd-MVAC group and 134 (55%) patients in the GC group. Gastrointestinal (GI) grade 3 disorders were more frequently observed in the dd-MVAC arm (p = 0.003), as well as asthenia of grade 3 (p < 0.001). It is worth noting that patients treated with both chemotherapies had 6.5%-6.9% cardiovascular toxicities and both 1.5–7.0 fold increase in long-term risk of CAD[5]. In addition, the GC regime is shorter – 4 cycles in comparison to 6 cycles.
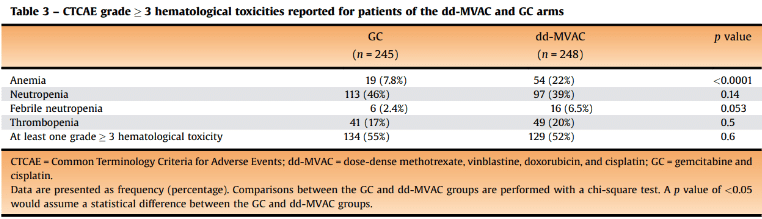
Carboplatin can be a relatively safe substitute for cisplatin in GC treatment regimes for patients unfit for cisplatin therapy – an article[6] published in the Journal of Clinical Oncology (Q1, impact factor 50.71) in 2009 presented the results of a randomized phase II/III trial assessing gemcitabine and carboplatin therapy vs Methotrexate/Carboplatin/Vinblastine (MCAVI) for 178 patients (median age 72) unfit for cisplatin therapy. According to the study results, there was no real significance in median OS or ORR, but gemcitabine and carboplatin had less serious (grade 3/4) toxicity overall in comparison to MCAVI (9 versus 21 percent), including neutropenia (52 versus 63 percent) and febrile neutropenia (5 versus 15 percent). However, it was associated with a higher rate of serious thrombocytopenia (47 versus 18 percent).
Immunotherapy:
Before moving to the details about the survival and QOL data of specific urothelial carcinoma treatments, it is important to evaluate the influence of Immune Checkpoint Inhibitors (ICIs) treatment on the elderly.
Geriatric patients with cancer can derive benefits from ICIs but have a lower threshold for and a higher rate of immune-related adverse events-related ICI discontinuation with increasing age – a brief report[7] published in JAMA Oncology (Q1, impact factor 31.777) in 2021 aimed to evaluate the clinical outcomes and spectrum of immune-related adverse events (irAEs) following treatment with single-agent immune checkpoint inhibitors in 928 patients (median age of 83).
The results show that among all 928 patients, 383 (41.3%) experienced ≥1 irAE(s), including 113 (12.2%) that were reported to be grade (G) 3 to 4 based on Common Terminology Criteria for Adverse Events (version 5.0). The median time to irAE onset was 9.8 weeks; 219 (57%) occurred within the first 3 months after ICI initiation. Discontinuation of treatment with ICIs owing to irAEs occurred in 137 (16.1%) patients. There was no significant difference in the rate of irAEs among patients aged younger than 85, 85 to 89, and 90 years or older.
The writers conclude that single-agent checkpoint inhibitors may be effective and generally well tolerated in patients older than 80 years, suggesting that age alone should not preclude patients from treatment with ICIs.Treating patients with ICIs is non-inferior and sometimes superior to other chemotherapy regimes in aspects of quality of life – a meta-analysis[8] published in The Journal of the National Cancer Institute (Q1, impact factor 13.506) in 2022 aimed to describe the change in QOL and symptomatology in patients receiving ICIs for cancer. The results show that Global QOL did not change over time in patients treated with ICIs (k = 26, n = 6974; P = .19). Larger improvements in global QOL was observed in patients receiving ICI vs non-ICI regimens (k = 16, ICI: n = 3588; non-ICI: n = 2948; P < .001). Physical functioning did not change in patients treated with ICIs (k = 14, n = 3169; P = .47); there were no differences in mean change between ICI vs non-ICI regimens (k = 11, n = 4630; P = .94). Regarding symptoms, appetite loss, insomnia, and pain severity decreased, but dyspnea severity increased in patients treated with ICIs (k = 14, n = 3243-3499; P < .001). Insomnia severity was higher in patients treated with ICIs than in non-ICI regimens (k = 11, n = 4791; P < .001).
The immunotherapy for urothelial carcinoma is mainly suggested for patients that are not eligible for Carboplatin or Cisplatin-based chemotherapies:
PD-L1 positive patients – pembrolizumab or atezolizumab are alternative choices for patients:
Pembrolizumab – has an extended median OS, with treatment-related AEs that can worsen the patient’s quality of life – an article[9] published in the Journal of Clinical Oncology in 2020 (Q1, impact factor 50.71) presented the results of a phase II study investigating long term outcomes of Pembrolizumab in 370 patients (Median age was 74 years) with locally advanced or metastatic urothelial cancer. According to the study results, in terms of survival, in patients with ECOG PS (Eastern Cooperative Oncology Group performance status) 2 and visceral metastatic disease, ORR was 22.3%, median OS was 7.8 months (95% CI, 5.1 to 10.6 months), and median DOR (duration of response) was 24.0 months (95% CI, 7.5 months to NR).
Regarding safety profile and QOL, the treatment has side effects that can influence the patient’s quality of life, such as fatigue (18.1%), pruritus (17.8%), rash (11.6%), decreased appetite (10.8%), diarrhea (9.2%) and nausea (8.6%). As a result, 16.5% of the patients had discontinued treatment because of AE. In terms of administration, the treatment is given IV, every 3 weeks for up to 24 months.Atezolizumab – also shows an extended median OS, with treatment-related AEs that can worsen the patient’s quality of life – an article[10] published in the Lancet journal in 2017 (Q1, impact factor 79.321) presented the results of a phase II single-arm study investigating long term outcomes of Atezolizumab in 119 patients (Median age was 73 years) with locally advanced or metastatic urothelial cancer. According to the study results, in terms of survival, the median overall survival was 6.2 months in those with 2 risk factors (ECOG PS >1 and baseline visceral metastasis).
Regarding safety and QOL, treatment-related adverse events observed in ≥10% of patients (any grade) were fatigue, diarrhea, and pruritus. Grade 3 or 4 treatment-related events occurred in 19 patients (16%), most frequently fatigue (3·4%, n=4), increased alanine aminotransferase (3·4%, n=4), and increased aspartate aminotransferase (2·5%, n=3). In terms of administration, the drug was given intravenously every 21 days until unacceptable toxicity or investigator-assessed radiographic progression.Other anti-PD-L1 drugs are available as second and third-line therapies, but their safety profile is quite similar to the two drugs we mentioned above.
Anti-FGFR therapy
FGFR2/3 mutations or FGFR3 fusions patients:
Erdafitinib – can be only given to patients with the genomic profile mentioned above and also shows side effects that influence QOL, but show some extension in survival and can be given orally – an article[11] published in The New England Journal of Medicine (Q1, impact factor 79.258) in 2019 presented the results of a phase II open-label study investigating long term outcomes of Erdafitinib in 99 patients (Median age was 68 years) with locally advanced or metastatic urothelial cancer. According to the study results, in terms of survival, the median overall survival was 13.8 months and progression-free survival was 5.5 months (with no intra-variable analysis of patients with distal metastasis). Patients with bone metastasis had a 48% rate of response (95% CI, 26-69), and those with lung metastasis had a 40% rate of response (95% CI, 28-53).
Regarding safety and QOL, common adverse events of grade 3 or higher were hyponatremia (11%), stomatitis (10%), and asthenia (7%), and patients also suffered from less severe side effects (grades 1-2) that can influence QOL like diarrhea, dry mouth, decreased appetite, fatigue, and dry skin. In terms of drug administration, Erdafitinib is delivered orally and given today in a tab of 8 mg once daily with a dose increase to 9 mg daily if criteria are met.
Palliative radiotherapy (RT):
Palliative radiotherapy in poor performance status[12] patients show worst survival in comparison to patients with good performance status – a retrospective multicenter study [13] published in the International Journal of Radiation Oncology*Biology*Physics journal (Q1, impact factor 7.038) in 2019 investigated the effectiveness of palliative pelvic radiation therapy (PRT) in 241 patients (median age was 80 years) with bladder cancer and identified factors associated with treatment outcome. The mOS from the last fraction of RT was 3.2 months in the stage IV and recurrence subgroup.
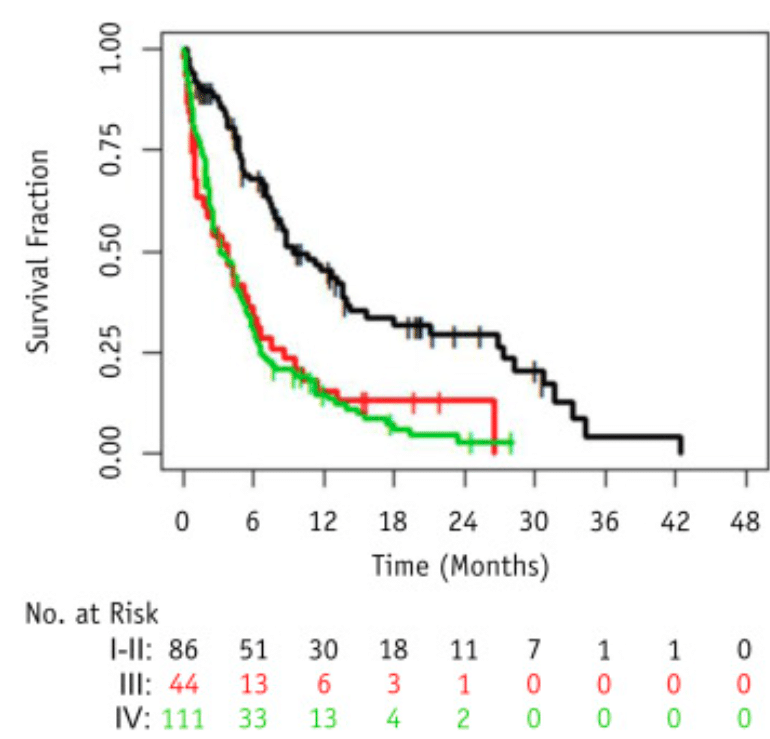
The writers suggest that a shorter RT regime may be more practical in poor performance status patients given the limited life expectancy for the majority of patients and the fact that it has no compromise in efficacy.
Palliative radiotherapy can help with local bladder symptom relief but have some worth mentioning toxicities – a systematic review[14] and meta-analysis published in Acta Oncologica journal (Q1, impact factor 4.31) in 2021 tried to determine if a higher dose of RT was associated with improved symptoms response rates. The researchers found one randomized, four prospective, and eight retrospective non-comparative observational studies including 1320 patients (most of which are older than 75 and had advanced or metastatic disease) who received palliative bladder radiotherapy for symptom relief (three studies included patients with both locally advanced disease and the remaining 10 studies included patients with both locally advanced and metastatic disease).
According to the research results, The pooled response rates for hematuria, dysuria, and frequency symptoms were 74%, 58%, and 71% respectively, depending on the radiation dose fraction rate. In the case of toxicities, grade 3 gastrointestinal and genitourinary toxicity occurred in up to 26% of patients, Regarding this matter, it is worth mentioning that most toxicities occurred in older studies (1991-2000), and the reviewed trials from 2013-2019 showed less toxicity, which may be contributed to advances in radiotherapy technologies and protocols.
Regarding the quality of life metric, only one study evaluated the quality of life data, showing no evidence of a difference in the change between the pretreatment and end-of-treatment assessment between the two arms (35 Gy/10 fractions vs. 21 Gy in 3 fractions).
- Are there any study protocols/trials or experimental treatments that would meet the above criteria?
It is important to note that most trials for experimental therapies require unresponsiveness or disease progression under more accepted therapies for metastatic disease as an inclusion criterion. In addition, we did not find any active trial that prioritizes QOL metrics instead of survival. We included three trials conducted in Canada that may be relevant to the patient, but because that are hundreds of active clinical trials for urothelial carcinoma in the US and Canada, we will need more information about the patient’s expanded medical history, medications, prognosis, tumor mutations, and personal preferences regarding location to see if the trials fit her clinical status and personal preferences.
Therapy Tested | Trial Description | Phase & Status | Locations and Contact Details |
RO7502175 as a Single Agent and in Combination With Atezolizumab | This is a first-in-human study to evaluate the safety, tolerability, pharmacokinetics (PK), and anti-tumor activity of RO7502175 when administered as a single agent and in combination with atezolizumab in adult participants with locally advanced or metastatic solid tumors. Participants will be enrolled in 2 stages: dose escalation and dose expansion. | Recruiting, Phase 1 | Location: Princess Margaret Cancer Centre, Toronto, Ontario, Canada, M5G 1Z5. Contact: Reference Study ID Number: GO43860 https://forpatients.roche.com/ Phone: 888-662-6728 (U.S. Only) |
Testing the Addition of Tazemetostat to the Immunotherapy Drug, Pembrolizumab (MK-3475), in Advanced Urothelial Carcinoma | This phase I/II trial studies the side effects and best dose of Tazemetostat and how well it works when given together with pembrolizumab in treating patients with urothelial metastatic carcinoma. Tazemetostat may stop the growth of tumor cells by blocking some of the enzymes needed for cell growth. | Recruiting, Phase 1/2 | Location: University Health Network-Princess Margaret Hospital, Toronto, Ontario, Canada, M5G 2M9 Contact: Phone: 416-946-4501 |
A Study of Disitamab Vedotin Alone and With Pembrolizumab in Urothelial Cancer That Expresses HER2 | This study is being done to see if a drug called disitamab vedotin, alone or with pembrolizumab, works to treat HER2 expressing urothelial cancer. It will also test how safe the drug is for participants. Participants will have cancer that has spread in the body near where it started (locally advanced) and cannot be removed (unresectable) or has spread through the body (metastatic). It will also study what side effects happen when participants get the drug. A side effect is anything a drug does to your body besides treating the disease. | Recruiting, Phase 2 | Location 1: British Columbia Cancer Agency – Vancouver Centre, Vancouver, British Columbia, Canada, V5Z4E6 Location 2: Jewish General Hospital Montreal, Quebec, Canada, H3T 1E2 Location 3: Centre hospitalier universitaire de Sherbrooke (CHUS), Sherbrooke, Québec, Canada, J1H 5N4 Phone: 866-333-7436 |
References
- Jodon G, Fischer SM, Kessler ER. Treatment of Urothelial Cancer in Elderly Patients: Focus on Immune Checkpoint Inhibitors. Drugs Aging. 2018;35(5):409-421. doi:10.1007/s40266-018-0540-8
- National Comprehensive Cancer Network. Bladder Cancer (Version 1.2023). https://www.nccn.org/professionals/physician_gls/pdf/bladder.pdf
- European Association of Urology. Upper Urinary Tract Urothelial Carcinoma 2O23. https://d56bochluxqnz.cloudfront.net/documents/full-guideline/EAU-Guidelines-on-Upper-Urinary-Tract-Urothelial-Carcinoma-2023.pdf
- Pfister C, Gravis G, Fléchon A, et al. Randomized Phase III Trial of Dose-dense Methotrexate, Vinblastine, Doxorubicin, and Cisplatin, or Gemcitabine and Cisplatin as Perioperative Chemotherapy for Patients with Muscle-invasive Bladder Cancer. Analysis of the GETUG/AFU V05 VESPER Trial Secondary Endpoints: Chemotherapy Toxicity and Pathological Responses. Eur Urol. 2021;79(2):214-221. doi:10.1016/j.eururo.2020.08.024
- Han XJ, Li JQ, Khannanova Z, Li Y. Optimal management of coronary artery disease in cancer patients. Chronic Dis Transl Med. 2020;5(4):221-233. Published 2020 Jan 14. doi:10.1016/j.cdtm.2019.12.007
- De Santis M, Bellmunt J, Mead G, et al. Randomized phase II/III trial assessing gemcitabine/ carboplatin and methotrexate/carboplatin/vinblastine in patients with advanced urothelial cancer “unfit” for cisplatin-based chemotherapy: phase II–results of EORTC study 30986. J Clin Oncol. 2009;27(33):5634-5639. doi:10.1200/JCO.2008.21.4924
- Nebhan CA, Cortellini A, Ma W, et al. Clinical Outcomes and Toxic Effects of Single-Agent Immune Checkpoint Inhibitors Among Patients Aged 80 Years or Older With Cancer: A Multicenter International Cohort Study. JAMA Oncol. 2021;7(12):1856-1861. doi:10.1001/jamaoncol.2021.4960
- Gonzalez BD, Eisel SL, Bowles KE, et al. Meta-Analysis of Quality of Life in Cancer Patients Treated With Immune Checkpoint Inhibitors. J Natl Cancer Inst. 2022;114(6):808-818. doi:10.1093/jnci/djab171
- Vuky J, Balar AV, Castellano D, et al. Long-Term Outcomes in KEYNOTE-052: Phase II Study Investigating First-Line Pembrolizumab in Cisplatin-Ineligible Patients With Locally Advanced or Metastatic Urothelial Cancer. J Clin Oncol. 2020;38(23):2658-2666. doi:10.1200/JCO.19.01213
- Balar AV, Galsky MD, Rosenberg JE, et al. Atezolizumab as first-line treatment in cisplatin-ineligible patients with locally advanced and metastatic urothelial carcinoma: a single-arm, multicentre, phase 2 trial [published correction appears in Lancet. 2017 Aug 26;390(10097):848]. Lancet. 2017;389(10064):67-76. doi:10.1016/S0140-6736(16)32455-2
- Loriot Y, Necchi A, Park SH, et al. Erdafitinib in Locally Advanced or Metastatic Urothelial Carcinoma. N Engl J Med. 2019;381(4):338-348. doi:10.1056/NEJMoa1817323
- Prigerson HG, Bao Y, Shah MA, et al. Chemotherapy Use, Performance Status, and Quality of Life at the End of Life. JAMA Oncol. 2015;1(6):778-784. doi:10.1001/jamaoncol.2015.2378
- Ali A, Song YP, Mehta S, et al. Palliative Radiation Therapy in Bladder Cancer-Importance of Patient Selection: A Retrospective Multicenter Study. Int J Radiat Oncol Biol Phys. 2019;105(2):389-393. doi:10.1016/j.ijrobp.2019.06.2541
- Tey J, Ho F, Koh WY, et al. Palliative radiotherapy for bladder cancer: a systematic review and meta-analysis. Acta Oncol. 2021;60(5):635-644. doi:10.1080/0284186X.2021.1880025
- Richters A, Boormans JL, van der Heijden MS, et al. Overall Survival of Patients Receiving Cisplatin or Carboplatin for Primary Metastatic Urothelial Carcinoma of the Bladder: A Contemporary Dutch Nationwide Cohort Study. Eur Urol Focus. 2022;8(4):995-1002. doi:10.1016/j.euf.2021.08.009
View as PDF


